order generic Lyrica Due to new information about the pathophysiology and biomechanics of degenerative lumbar spine disease, the surgical treatment of this disease has undergone a significant increase over the past forty years.
Novel diagnostic approaches and the development of new materials provided the impetus to produce new types of instrumentation, and these instruments have led to the modernization of interbody fusion including PLIF, TLIF and ALIF methods. These interventions are currently performed in either an open mini-invasive or endoscopic manner. The open interventions are indicated in cases where the spinal canal stenosis is caused by severe degenerative lesions affecting the motion of intervertebral discs, joints, ligaments, or vertebral arch. Despite the development of other surgical techniques (e.g., functional disc substitutes, dynamic stabilization), the posterior interbody fusion represents a powerful approach in the surgical treatment of degenerative stenosis of the spinal canal.
Pīshvā The PLIF method was first applied in the 1940s by Briggs and Milligan who inserted crushed bone grafts into the intervertebral space, and the bone grafts insertion technique was further developed by Cloward (Cloward, 1953). Due to complications associated with autografts (i.e., pain at the sampling site, procedure prolongation, etc.), the PLIF surgical technique was improved in the 1980s, and new implants constructed of various materials were developed (Bienik and Swiecki, 1991; Brantigan et al, 1994; Khoo et al, 2002; Sramek et al, 2010). Likewise, novel diagnostic tools have been developed including MRI, 3D CT, SPECT-CT (Crock, 1976, Modic et al, 1988; Blumenthal et al, 1988), and new materials (e.g., ceramic, titanium, PEEK) have yielded new types of implants leading to the modernization of the interbody fusion via PLIF techniques (Alexander et al, 2002; Bessho et al, 1997; Brayan et al, 2002; Ciappetta et al, 1997; Kokubo, 1990; Yamamuro, 1995; Hashimoto et al, 2002; Thalgott et al, 2002; Sandhu, 2003). Currently, the majority of implants for PLIF consist of two separate components, including the solid cage shape and osseoconductive material (i.e., TCP, BMP) that ensures osteoblastic activity and the interbody fusion formation. To date, no material with both suitable mechanic properties and high grade bioactivity is currently available. For instance, solid materials (e.g., medical steel, titanium, PEEK) lack bioactivity that is able to support the osseoconduction (Carlson et al, 1988; Williams and McNamara, 1987; Zdeblick and Philips, 2003). Likewise, bioactive or resorbable materials (e.g., glass-ceramic, hydroxyapatite, polysacharides) do not meet the mechanical requirements for fusion implants of intervertebral discs (Hench el al, 1971; Filip et al, 1996; Sobale et al, 1990; McAfee, 1986).
Currently, the majority of implants consist of cages that form various shapes. The perimeter is constructed from a solid material that ensures the structural strength. The centre of the cage is hollow and is filled by bone grafts or osseoconductive material (e.g. bi- or tri-calcium phosphate) to promote bone fusion in this part of the implant. The optimal implant for an interbody fusion should imitate the properties of the bone tissue by combining sufficient mechanical strength as well as bioactive surface. Therefore, the mechanical strength and the shape of the implant should ensure the primary stability of the segment of the lumbar spine following the operation. Furthermore, the bioactive surface should allow stimulation of osteoblast proliferation at the interface of the implant and bone, and should promote activation of their migration along the implant surface. The bioactive surface should also act as a conductor for osteoblast migration to the fixed vertebral bodies to form the fusion. This quality would prevent the requirement for additional filling of the implant by osseoconductive material. The aim of our work was to create an implant with optimal strength and bioactivity in an attempt to replace the use of autografts and two-compartment implants for PLIF.
When the conservative treatment fails, patients from categories LS syndrome and FBS syndrome (Failed Back Surgery syndrome) are often referred to surgical management via posterior interbody fusion (Benzel et al, 2003; Cloward, 1953; Crock, 1976; Daniaux, 1986; Dove, 1990; Gurr et al, 1999; Cho et al, 2002). The indication for this surgery is based on neurological finding. In general, the patient predominantly either suffers from back pain associated with progression of root lesion in a lower extremity or with neurogenic claudications in the lower limbs, and shows no reaction to the full conservative therapy algorhytm (Anderson, 2000; Brinnckman et al, 1989; Brodke et al, 1997; Cloward, 1953, Hrabalek et al, 2009; Palecek et al, 1994; Fischgrund et al, 1997). Furthermore, the disease is supported by graphic images of compression of the neural structures caused by degenerative lesions (Knudson, 1944; Crock, 1978; Modick et al, 1983; Sonntag and Theodore, 2000). The desired clinical effect can be achieved by the decompression of neural structures together with spondylodesis of the affected spine segment using PLIF (Steffee, 1988; Hashimoto el al, 2002; Dick, 1987; Wang et al, 2005). When surgical treatment is necessary, no acceptable scientific long-term evidence of efficacy exists for any type of surgical treatment of the degenerative lumbar spine disease (Brodke et al, 1997; Benzel, 2003; Sonntag and Theodore, 2000; Palecek et al, 1994).
We performed PLIF using autografts that were developed in the 1980s. An autograft (mostly iliac crest bone grafts) stripped of connective tissue was inserted under compression into the intervertebral space. The best stability was achieved by transpedicular fixation of the operated segment necessary for osteointegration of the grafts via their remodeling and PLIF formation (Bauer and Muschler, 2000). The advantage of a recently collected autolog bone graft has been the presence of live bone cells with mineralized extracellular matrix. The biological activity, structure and proteins of bone morphogenesis are important prerequisites of the fusion. In addition, clinical experiences from the first half of the 20th century have proved better surgical outcomes with autolog grafts in comparison to simple decompression (Cloward, 1953; Dawson et al, 1981; Dick, 1987; Carlson et al, 1988). Autolog grafts in this form have been the gold standard for PLIF in the majority of spondylosurgical clinics through the end of the 20th century. Despite improving surgical outcomes with a growing number of operated patients, new complications still exist regarding this otherwise successful surgical technique (Kurz et al, 1989).
The most common complications associated with this surgery include problems with bone graft sampling in that limitations are present in bone size and structure that may be safely collected from a live patient in cases of extensive intervention. Furthermore, patients can suffer from unpleasant reactions including debilitating postoperative pain, infection, seroma, cosmetic defects, nerve injury, hip fractures, vessel injury and blood loss. These adverse reactions can occur in 10 to 39% of cases (Arrington et al, 1996; Banwart el al, 1994; Banwart et al, 1995). Therefore, these reactions and other problems have led to search for artificial materials for PLIF. The optimal material for PLIF substituting bone grafts should ideally have the following characteristics. First, the material should show solid structural support (load resistance immediately after implantation). Second, the material should display osseoconductivity and bioactivity or the ability to bind with a bone, fusion support without any other additional material (e.g., bone, BCP etc.). Third, the material should provide the possibility for a radiographical assessment of the bone fusion process. Finally, the material should show biomechanic properties (elasticity modulus similar to bone).
The most available implants were constructed of medical steel (Bagby, 1988). However, these implants did not meet our notion of sufficient strength accompanied by bioactivity. Spondylosurgeons in Charkov (Professor Gruntovskij) have successfully used corundum implants in combination with hydroxyapatite for PLIF in the surgical treatment of degenerative lumbar spine disease in the 1980s. According to results of this clinic, the success of this implant resulted from its prism shape with projections firmly anchored in the intervertebral space that helped the implant to fixate the segment with or without transpedicular fixation following operation. Due to its bioinertion, hydroxyapatite was added, and this soft material was placed around the corundum (Rowlings, 1993; Gogolewski et al, 1993). Therefore, this implant stimulated formation of osteoblasts, and served as a conductor for their migration between adjacent surfaces of adjoining vertebral bodies. In the early 1990s, another type of prosthesis produced from bioactive glass-ceramic was developed by Electric Nippon Glass, and was used by Japanese orthopedists for PLIF (Yamamuro, 1995, Kokubo, 1990). While transpedicular fixation was added to PLIF due to its fragility, the bioactivity of the implant surface allowed fusion due to migration of bone cells along its surface without addition of any supporting material (e.g., bone, hydroxyapatite) (Sobale, 1990; Yamamuro, 1995). Based on these experiences, we began searching for a material for PLIF implant that would combine the advantages of both the shape and the strength of corundum and the bioactivity of glass-ceramic used in the early 1990s. Thereby, the combination of these two properties would allow strong anchoring of this material in the intervertebral space, the restoration of anatomy in the operated segment, the stabilization of unstable segment, and the formation of interbody fusion associated with osseoconductive properties without addition of another material and without the risk of migration.
Unlike bioinert or biotolerant materials, bioactive glass-ceramic material BAS-O, forms a strong chemical bond with live bone tissue (Fatley et al, 1979; Urban, 1992). Material BAS-O is prepared by progressive steps, such as sintering, controlled crystallization and others. The controlled crystallization allows control of processes that determine the bioactive ability of the final material including material transformation, the control of chemical structure, and the structure of the glass phase (Strnad, 1992). The ability of this material to form a strong bond with bone tissue results from the formation of an apatite layer on the material surface resulting in the connection of the bioactive material with body fluid. Crystallographic chemical characteristic of apatite released on the material surface is similar to the organic part of the bone tissue. Thereby, the stability of the operated segment without micromovements and the tight contact of the material without microgaps are necessary for perfect chemical bond BAS-O / live tissue. Otherwise, a risk of connective tissue penetration exists that can prevent the chemical bond on the bone / implant interface (Kokubo, 1990; Urban, 1992).
The most important finding for the planned use of the lumbar implant necessitated that the biochemical and mechanical properties of the glass-ceramic BAS-O mimic the cortical bone tissue. According to the Young model, the shape of their implant exceeded twice the strength of the vertical load, and was close to its flexural strength. Therefore, we based our implant shape on our previous experiences and according to the models that we observed during our study visits. Together with size and shape development, we also created application instrumentation used for the intervertebral space as well as the operation procedure. At this time, the fragility of the ceramic in the contact with steel represented our only disadvantage in that this fragility could cause problems with insertion using metal application instrumentation. The application instrumentation was coated by Teflon in order to prevent damage to the implant. A rectangular prism-shaped implant (25 mm long, 8 mm high and 10 mm wide) was progressively developed after repeated experiments with cadavers from 1991 to 1993 (Filip et al, 1995). “Winglets” have been placed on the opposite sides of the prism (Figure 1).
The winglets cut into the adjacent vertebral bodies after its rotation by 90 degrees, and the implant was firmly attached within the space without a risk of migration into the spinal canal. Due to its bioactivity, the implant should stimulate migration of bone cells along its surface to form interbody fusion. The application technique for the glass-ceramic implant was the same as with other implants for PLIF. During experimental application in cadavers, the implants were well-anchored in the vertebral bodies without compression of dural sac in the spinal canal, and this placement was confirmed by imaging techniques (X-ray and CT).
Implaspin in clinical practice
Encouraged by these experimental results, we began to use this type of implant in clinical practice in indications for PLIF instead of the glass-ceramic implant since 2002 (Figure 9).
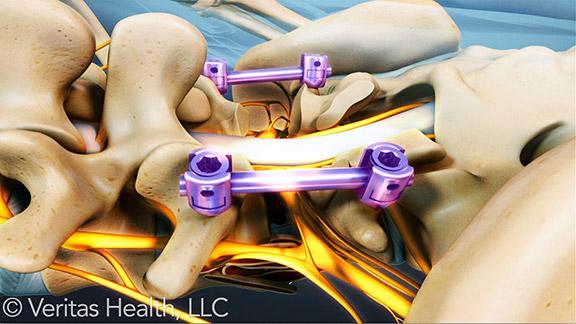
The operation technique PLIF was identical to the operation technique used in cadavers (Filip et al, 2010). For example, we decompressed the nervous structures through posterior median line approach, and we then radically removed the degenerated intervertebral disc under the control of the operation microscope. Afterwards, we removed the surfaces of the adjacent vertebral bodies, and we then inserted the bioactive titanium implant using the innovated instrumentation (Figure 10). Finally, we added transpedicular fixation of the whole segment (Synthes, Signus, Easy spine, etc.) (Figure 11).

Fig. 9. Implant from bioactive titanium – Implaspin (2002).

Fig. 10. Insertion of Implaspin into the interbody space.

Fig. 11. Transpedicular application of screws (SIGNUS)
To date, we have not observed any complications associated with the implant application into the interbody space. According to the postoperative scans, the implant was always placed in the correct position with winglet penetration into the spongious tissue of the adjacent vertebral bodies. We have selected the size empirically according to the extent of osteochondrosis of the affected disc and the degenerative lesions of the surrounding tissues on scans (X-ray, CT, MRI) during the intervention. In the majority of cases, we used implants (8 or 10 mm high) with angle 4% to maintain lordosis in the lumbar area (Figure 12).

Fig. 12. Fixation L4/5 (Implaspin plus transpedicular screws Signus).
أضف تعليقك